Fabrication and Release Properties of Naproxen-loaded Dioscorea dumetorum (Bitter yam) Starch Biodegradable Nanoparticles
DOI:
https://doi.org/10.63224/tnm.2025.1.001Keywords:
Starch nanoparticles, Carboxymethylated starch nanoparticles, Dioscorea dumetorum, Naproxen, Drug release studiesAbstract
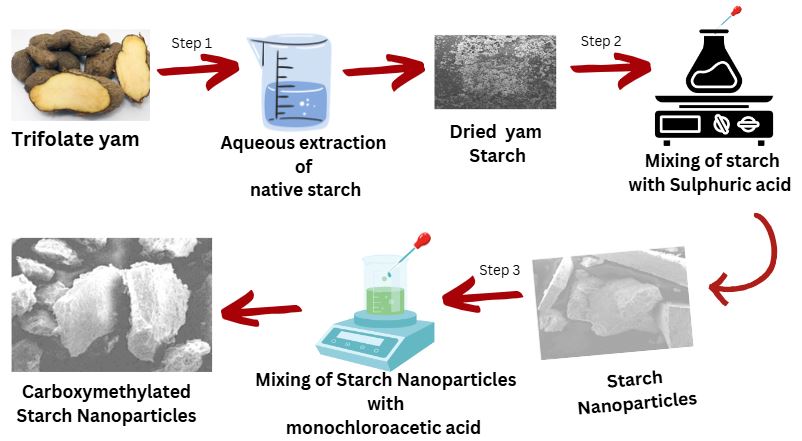
The growing demand for polymers to improve drug delivery profiles (particularly for poorly aqueous soluble drugs) has prompted a global search for new polymers. The drug loading, dissolution, and release characteristics of trifoliate yam (Dioscorea dumetorum) starch, a biodegradable polymer, were evaluated using naproxen as the model drug. Trifoliate starch nanoparticles (TSNP) were created by acid hydrolysis of trifoliate native starch (TNS) with H2SO4. In contrast, carboxymethylated trifoliate starch nanoparticles (CTSNP) were created by reacting the TSNP with sodium hydroxide and monochloroacetic acid. Scanning electron microscopy analysis revealed that TSNP were formed by acid hydrolysis. Modification of the TSNP resulted in a significant particle size reduction (86.5 ± 5.60 nm) and changes in the morphology of these starch forms compared to the native trifoliate starch (1616.0 ± 0.54 nm). The addition of a carboxylate functional group to the TSNP molecule as a means of modification was confirmed by significant peaks in the Fourier transform Infrared spectroscopy’s 1600-1300 cm-1 absorption band region. All of the starch forms, TNS, TNSP and CTNSP demonstrated significant drug loading capacity and efficiency (86.7%, 86.4%, and 84.9%), respectively. The TNS demonstrated delayed naproxen release, whereas the TSNP and CTSNP demonstrated a biphasic release profile (immediate and sustained release). Naproxen release kinetics from most formulations followed the Korsmeyer-Peppas model of non-Fickian diffusion through the polymeric matrix. A loading time of one hour was found to optimize loading efficiency. The starch nanocarriers developed in this study can be used as vehicles to enhance, control, and prolong the release of poorly soluble drugs.
References
Andonova V. Synthetic polymer-based nanoparticles: intelligent drug delivery systems. In: Acrylic Polymers in Healthcare, Reddy B (Ed.); IntechOpen: India; 2017; 101–125. DOI: https://doi.org/10.5772/intechopen.69056.
Ajala TO, Omoteso OA, Awe OM. The Design and Evaluation of Ciprofloxacin-Loaded Nanoformulations Using Ipomoea Batatas Starch Nanoparticles. Futur J Pharm Sci. 2023;9(1):37. DOI: https://doi.org/10.1186/s43094-023-00487-z.
Odeniyi MA, Omoteso OA, Adepoju AO, Jaiyeoba KT. Starch Nanoparticles in Drug Delivery: A Review. Polim. Med. 2019;48(1):41–45. DOI: https://doi.org/10.17219/pim/99993.
Sur S, Rathore A, Dave V, Reddy KR, Chouhan RS, Sadhu V. Recent Developments in Functionalized Polymer Nanoparticles for Efficient Drug Delivery System. Nano-Struct Nano-Objects. 2019;20:100397. DOI: https://doi.org/10.1016/j.nanoso.2019.100397.
Bamiro OA, Odeniyi MA, Addo RT. Native and Modified Oryza glaberrima Steud Starch Nanocrystals: Solid-state characterization and Anti-tumour drug release studies. Br J Pharm. 2021;6(1):790. DOI: http://doi.org/10.5920/bjpharm.790.
Odeniyi MA, Adepoju AO, Jaiyeoba KT. Native and modified Digitaria exilis starch nanoparticles as a carrier system for the controlled release of naproxen. Starch‐Stärke. 2019;71(9-10):1900067. DOI: https://doi.org/10.1002/star.201900067.
Garcia MA, Cesar AL, Garcia CF, Faraco AA. Starch applied for pharmaceutical use. In: Starch Industries: Processes and Innovative Products in Food and Non-Food Uses, 1st ed.; Cereda MP, Vilpoux OF (Eds.); Academic Press: USA; volume 3, 16, 2024; 349-376. DOI: https://doi.org/10.1016/B978-0-323-90842-9.00004-2.
Omoteso OA, Adebisi AO, Kaialy W, Asare-Addo K, Odeniyi MA. Effect of pregelatinization and carboxymethylation on starches from African rice and Fonio: Influence on release of low melting-point drug. Br J Pharm. 2019;4(2):4-11. DOI: https://doi.org/10.5920/bjpharm.645.
Omoteso OA, Adebisi AO, Odeniyi MA. Impact of Thermal and Chemical Modifications on the Compression and Release Properties of Bambara Nut Starches in Directly Compressed Tablet Formulations. Starch‐Stärke. 2018;70(11–12):1700308. DOI: https://doi.org/10.1002/star.201700308.
Odeniyi M, Omoteso OA, Adebisi AO. Solid state characterization and rheological properties of native and modified Bambara groundnut (Vigna subterranean) starches. J Excip Food Chem. 2017;8(3):42-51.
Kumari B, Sit N. Comprehensive review on single and dual modification of starch: Methods, properties and applications. Int J Biol Macromol. 2023;253(3):126952. DOI: https://doi.org/10.1016/j.ijbiomac.2023.126952.
Garcia MA, Garcia CF, Faraco AA. Pharmaceutical and biomedical applications of native and modified starch: a review. Starch‐Stärke. 2020;72(7-8):1900270. DOI: https://doi.org/10.1002/star.201900270.
Liu Q, Cai W, Zhen T, Ji N, Dai L, Xiong L, Sun Q. Preparation of debranched starch nanoparticles by ionic gelation for encapsulation of epigallocatechin gallate. Int J Biol Macromol. 2020;161:481-491. DOI: https://doi.org/10.1016/j.ijbiomac.2020.06.070.
Torres FG, De-la-Torre GE. Synthesis, characteristics, and applications of modified starch nanoparticles: A review. Int J Biol Macromol. 2022;194:289-305. DOI: https://doi.org/10.1016/j.ijbiomac.2021.11.187.
Marta H, Rizki DI, Mardawati E, Djali M, Mohammad M, Cahyana Y. Starch nanoparticles: Preparation, properties and applications. Polymers. 2023;15(5):1167. DOI: https://doi.org/10.3390/polym15051167.
Chavan P, Sinhmar A, Nehra M, Thory R, Pathera AK, Sundarraj AA, Nain V. Impact on various properties of native starch after synthesis of starch nanoparticles: A review. Food Chem. 2021;364:130416. DOI: https://doi.org/10.1016/j.foodchem.2021.130416.
Campelo PH, Sant’Ana AS, Clerici MT. Starch nanoparticles: production methods, structure, and properties for food applications. Curr. Opin. Food Sci. 2020;33:136-140. DOI: https://doi.org/10.1016/j.cofs.2020.04.007.
Li C, Guo Y, Chen M, Wang S, Gong H, Zuo J, Zhang J, Dai L. Recent preparation, modification and application progress of starch nanocrystals: A review. Int J Biol Macromol. 2023:126122. DOI: https://doi.org/10.1016/j.ijbiomac.2023.126122.
Qin Y, Xue L, Hu Y, Qiu C, Jin Z, Xu X, Wang J. Green fabrication and characterization of debranched starch nanoparticles via ultrasonication combined with recrystallization. Ultrason Sonochem.2020;66:105074. DOI: https://doi.org/10.1016/j.ultsonch.2020.105074.
Lin Z, Cheng H, He K, McClements DJ, Jin Z, Xu Z, Meng M, Peng X, Chen L. Recent progress in the hydrophobic modification of starch-based films. Food Hydrocoll. 2024;151:109860. DOI: https://doi.org/10.1016/j.foodhyd.2024.109860.
Cazotti JC, Fritz AT, Garcia‐Valdez O, Smeets NM, Dubé MA, Cunningham MF. Graft modification of starch nanoparticles with pH‐responsive polymers via nitroxide‐mediated polymerization. J Polym Sci. 2020;58(16):2211-2220.DOI: https://doi.org/10.1002/pol.20200337.
Wang Y, Hu Q, Li T, Ma P, Zhang S, Du M, Chen M, Zhang H, Dong W. Core–shell starch nanoparticles and their toughening of polylactide. Ind Eng Chem Res. 2018;57(39):13048-13054. DOI: https://doi.org/10.1021/acs.iecr.8b02695.
Kumari S, Yadav BS, Yadav RB. Synthesis and modification approaches for starch nanoparticles for their emerging food industrial applications: A review. Food Res Int. 2020;128:108765. DOI: https://doi.org/10.1016/j.foodres.2019.108765.
Chang R, Ji N, Li M, Qiu L, Sun C, Bian X, Qiu H, Xiong L, Sun Q. Green preparation and characterization of starch nanoparticles using a vacuum cold plasma process combined with ultrasonication treatment. Ultrason Sonochem. 2019;58:104660. DOI: https://doi.org/10.1016/j.ultsonch.2019.104660.
Operti MC, Bernhardt A, Grimm S, Engel A, Figdor CG, Tagit O. PLGA-based nanomedicines manufacturing: Technologies overview and challenges in industrial scale-up. Int J Pharm. 2021;605:120807. DOI: https://doi.org/10.1016/j.ijpharm.2021.120807.
Obioma OG, Doshima IB, Kwagh-Hal IJ. Potentials of yams’ utilizations in food, medicine and allied fields. IJCSPUB-Int. J. Curr. Sci (IJCSPUB). 2023;13(2):89-106.
Kumar A, Mishra S, Kumar S. Dioscorea dumetorum (Dioscoreaceae). In: Yam; Manjula et al; APRF: India; 2022; 1-8. DOI: https://doi.org/10.5281/zenodo.7147528.
Adetunji OA, Odeniyi MA, Itiola OA. Compression, mechanical and release properties of chloroquine phosphate tablets containing corn and trifoliate yam starches as binders. Trop. J Pharm Res. 2006;5(2):589-596.DOI: https://doi.org/10.4314/tjpr.v5i2.14636.
Obidiegwu JE, Lyons JB, Chilaka CA. The Dioscorea Genus (Yam)—An appraisal of nutritional and therapeutic potentials. Foods. 2020;9(9):1304. DOI: https://doi.org/10.3390/foods9091304.
Okereke GO, Igbabul BD, Ikya JK, Araka O. Physicochemical and functional properties of modified starches of white yam, trifoliate yam and sweet potato. J Food Chem Nanotechnol. 2022;8(2):50-60. DOI: https://doi.org/10.17756/jfcn.2022-126.
Weisman S. Naproxen for post-operative pain. J Pharm Pharm Sci. 2021;24:62-70. DOI: https://doi.org/10.18433/jpps31629.
Machado GC, Abdel-Shaheed C, Underwood M, Day RO. Non-steroidal anti-inflammatory drugs (NSAIDs) for musculoskeletal pain. Bmj. 2021;372:n104. DOI: https://doi.org/10.1136/bmj.n104.
Ribeiro H, Rodrigues I, Napoleão L, Lira L, Marques D, Veríssimo M, Andrade JP, Dourado M. Non-steroidal anti-inflammatory drugs (NSAIDs), pain and aging: Adjusting prescription to patient features. Biomed. Pharmacother. 2022;150:112958. DOI: https://doi.org/10.1016/j.biopha.2022.112958.
Tsume Y, Mudie DM, Langguth P, Amidon GE, Amidon GL. The Biopharmaceutics Classification System: subclasses for in vivo predictive dissolution (IPD) methodology and IVIVC. Eur J Pharm Sci. 2014;57:152-163. DOI: https://doi.org/10.1016/j.ejps.2014.01.009.
Huang FY, Wu XJ, Yu Y, Lu YH. Preparation and properties of cellulose laurate (CL)/starch nanocrystals acetate (SNA) bio-nanocomposites. Polymers. 2015;7(7):1331-1345. DOI: https://doi.org/10.3390/polym7071331.
Kittipongpatana OS, Sirithunyalug J, Laenger R. Preparation and physicochemical properties of sodium carboxymethyl mungbean starches. Carbohydr. Polym. 2006;63(1):105-112. DOI: https://doi.org/10.1016/j.carbpol.2005.08.024.
Stojanović Ž, Jeremić K, Jovanović S, Lechner MD. A comparison of some methods for the determination of the degree of substitution of carboxymethyl starch. Starch‐Stärke. 2005;57(2):79-83. DOI: https://doi.org/10.1002/star.200400342.
Xiao H, Yang T, Lin Q, Liu GQ, Zhang L, Yu F, Chen Y. Acetylated starch nanocrystals: preparation and antitumor drug delivery study. Int J. Biol Macromol. 2016;89:456-464. DOI: https://doi.org/10.1016/j.ijbiomac.2016.04.037.
Adetunji AO, Odeniyi MA, Itiola OA. Characterisation and controlled release properties of Entandophragma angolense gum in ibuprofen matrix tablets. Farmacia. 2015;63(1):57-64.
Angellier H, Choisnard L, Molina-Boisseau S, Ozil P, Dufresne A. Optimization of the preparation of aqueous suspensions of waxy maize starch nanocrystals using a response surface methodology. Biomacromol. 2004;5(4):1545-1551. DOI: https://doi.org/10.1021/bm049914u.
Lawal OS, Lechner MD, Kulicke WM. The synthesis conditions, characterizations and thermal degradation studies of an etherified starch from an unconventional source. Polym Degrad Stab. 2008;93(8):1520-1528. DOI: https://doi.org/10.1016/j.polymdegradstab.2008.05.010.
Chang R, Tian Y, Yu Z, Sun C, Jin Z. Preparation and characterization of zwitterionic functionalized starch nanoparticles. Int J Biol Macromol. 2020;142:395-403. DOI: https://doi.org/10.1016/j.ijbiomac.2019.09.110.
Spychaj T, Wilpiszewska K, Zdanowicz M. Medium and high substituted carboxymethyl starch: synthesis, characterization and application. Starch‐Stärke. 2013;65(1‐2):22-33. DOI: https://doi.org/10.1002/star.201200159.
Korsmeyer RW, Gurny R, Doelker E, Buri P, Peppas NA. Mechanisms of solute release from porous hydrophilic polymers. Int J. Pharm. 1983;15(1):25-35. DOI: https://doi.org/10.1016/0378-5173(83)90064-9.
Angellier H, Putaux JL, Molina‐Boisseau S, Dupeyre D, Dufresne A. Starch nanocrystal fillers in an acrylic polymer matrix. Macromolecular Symposia. 2005;221(1):349-376. DOI: https://doi.org/10.1002/masy.200550310.
Pooresmaeil M, Namazi H. Developments on carboxymethyl starch-based smart systems as promising drug carriers: A review. Carbohydr Polym. 2021;258:117654. DOI: https://doi.org/10.1016/j.carbpol.2021.117654.
Kittipongpatana OS, Chaitep W, Charumanee S, Kittipongpatana N. Effects of amylose content on the physicochemical properties of sodium carboxymethyl rice starches. CMU J Nat Sci. 2006;5:199-207.
Yanli W, Wenyuan G, Xia L. Carboxymethyl Chinese yam starch: synthesis, characterization, and influence of reaction parameters. Carbohydr. Res. 2009;344(13):1764-1769. DOI: https://doi.org/10.1016/j.carres.2009.06.014.
Li X, Gao WY, Huang LJ, Wang YL, Huang LQ, Liu CX. Preparation and physicochemical properties of carboxymethyl Fritillaria ussuriensis Maxim. starches. Carbohydr Polym. 2010;80(3):768-773. DOI: https://doi.org/10.1016/j.carbpol.2009.12.025.
Gallant DJ, Bouchet B, Baldwin PM. Microscopy of starch: evidence of a new level of granule organization. Carbohydr. Polym. 1997;32(3-4):177-191. DOI: https://doi.org/10.1016/S0144-8617(97)00008-8.
Kittipongpatana OS, Kittipongpatana N. Preparation and physicochemical properties of modified jackfruit starches. LWT-Food Sci. Technol. 2011;44(8):1766-1773. DOI: https://doi.org/10.1016/j.lwt.2011.03.023.
Yoshida T, Lai TC, Kwon GS, Sako K. pH-and ion-sensitive polymers for drug delivery. Expert Opin Drug Deliv. 2013;10(11):1497-1513. DOI: https://doi.org/10.1517/17425247.2013.821978.
Song S, Wang C, Pan Z, Wang X. Preparation and characterization of amphiphilic starch nanocrystals. J App. Polym. Sci. 2008;107(1):418-422. DOI: https://doi.org/10.1002/app.27076.
Govender T, Riley T, Ehtezazi T, Garnett MC, Stolnik S, Illum L, Davis SS. Defining the drug incorporation properties of PLA–PEG nanoparticles. Int J Pharm. 2000;199(1):95-110. DOI: https://doi.org/10.1016/S0378-5173(00)00375-6.
Getahun P, Gebre-Mariam T, Neubert HH. Fabrication and Evaluation of Cassava Starch Acetate Nanoparticles Loaded with Drugs of Various BCS Classes: Influence of Drug Solubility and Partition Coefficient. J Nanomater Mol Nanotechnol. 2019;11:2. DOI: 10.4172/2324-8777.1000259.
Saharan V, Kukkar V, Kataria M, Gera M, Choudhury PK. Dissolution enhancement of drugs. Part I: technologies and effect of carriers. Int J Health Res. 2009;2(2):107-124. DOI: https://doi.org/10.4314/ijhr.v2i2.55401.
Kaziem AE, Yang L, Lin Y, Tan Y, Song Z, Xu H, Zhang Z. Pest invasion-responsive hollow mesoporous silica-linked carboxymethyl starch nanoparticles for smart abamectin delivery. ACS Appl. Nano Mater. 2022;5(3):3458-3469. DOI: https://doi.org/10.1021/acsanm.1c04086.
Kotla NG, Gulati M, Singh SK, Shivapooja A. Facts, fallacies and future of dissolution testing of polysaccharide based colon-specific drug delivery. J Control Release. 2014;28;178:55-62. DOI: https://doi.org/10.1016/j.jconrel.2014.01.010.
Worsøe J, Fynne L, Gregersen T, Schlageter V, Christensen LA, Dahlerup JF, Rijkhoff NJ, Laurberg S, Krogh K. Gastric transit and small intestinal transit time and motility assessed by a magnet tracking system. BMC gastroenterol. 2011;11:145. DOI: https://doi.org/10.1186/1471-230X-11-145.
Kimura T, Higaki K. Gastrointestinal transit and drug absorption. Biol Pharm Bull. 2002;25(2):149-164. DOI: https://doi.org/10.1248/bpb.25.149.
Askarizadeh M, Esfandiari N, Honarvar B, Sajadian SA, Azdarpour A. Kinetic modeling to explain the release of medicine from drug delivery systems. ChemBioEng Rev. 2023;10(6):1006-1049. DOI: https://doi.org/10.1002/cben.202300027.
Ahmed L, Atif R, Eldeen TS, Yahya I, Omara A, Eltayeb M. Study the using of nanoparticles as drug delivery system based on mathematical models for controlled release. IJLTEMAS. 2019;8:52-56.
Published
Issue
Section
License
Copyright (c) 2025 Mr Adewale O. Adepoju, Ms. Vivian Okumah, Dr. Omobolanle A. Omoteso, Prof. Kolawole T. Jaiyeoba, Prof. Michael A. Odeniyi

This work is licensed under a Creative Commons Attribution 4.0 International License.
Under the CC BY 4.0 license, copyrights are retained by the authors, and anyone can have free and unlimited access, reuse, read and download any article for free. No permission is needed to reuse any part of articles published by Trends in NanoMed (TNM), including figures and tables. However, the original source must be clearly cited.

